Non-Bonding Electrons & Multiple Bonds
Non-bonding electrons
Non-bonding electrons affect the bond angle in molecules containing them.
Example:CH4, NH3 and H2O, all have the same electron-domain geometry; tetrahedral with different bond angles.
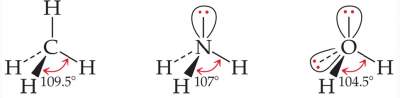
This is attributed to the repulsion between the high electron density in the non-bonding pairs and the close bonding pairs.
This results in different molecular geometries of these molecules.
Multiple Bonds
Multiple Bonds also affect bond angles in molecules containing them.
Example:Cl2CO molecule
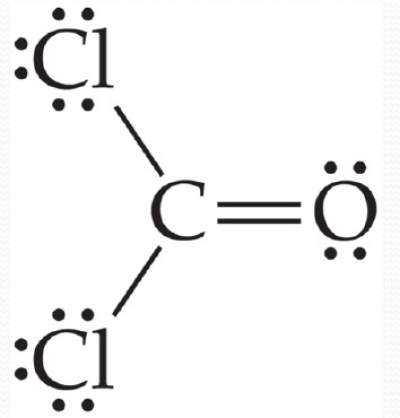
The central atom is surrounded by 3 bonding electron domains, hence both the electron-domain and molecular geometries should be:
Trigonal Planar with a bond angle of 120°
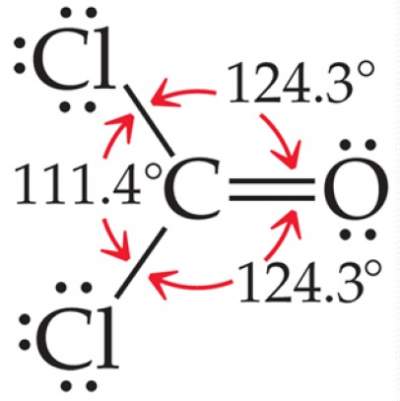
But, due to repulsion between the high electron density of the double bonds and the surrounding bonding electrons,
bond angles are distorted to minimize this repulsion.
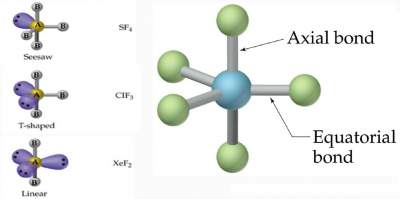
Remark
In case the number of electron domains around the central atom is five (5), where the electron domain geometry is trigonal bipyramidal,
nonbonding pairs are always placed in the equatorial plane to minimize repulsion.
For more details, please contact me here.




