Introduction to Limiting Reactant
Introduction
Example 1
2H2 + O2 → 2H2O
Each 2 moles of 2H2 need ONE mole of O2 to form 2 moles of H2 O
Assune we have 10 moles of H2, then we can ask the following:
How many moles of O2 are needed?
Solution:
5 moles
If we have only 10 mol of H2, 4 moles of O2.
Only 8 moles of H2 will be used leaving
2 unreacted(excess) H2 moles.
Meaning that the amount of O2 is limited of H2 needed
→ O2 is called "Limiting Reactant"
→ H2 is called "Excess Reactant"
Hence, at the end of the reaction:
- The limiting reactant will be completely consumed,
- Part of the excess reactant will remain after reaction
Important Remarks - 1
In a "limiting reactant case", a limiting reactant is used to calculate the number of moles of the product since the limiting reactant is totally consumed in the reaction.
Typically, In a "limiting reactant case", the main task is to find the limiting reactant, then the excess reactant.
Sometimes, finding the excess reactant is easier and will lead to the limiting reactant.
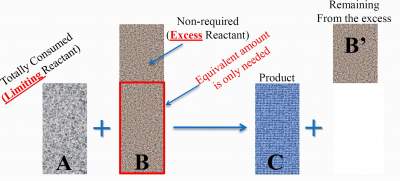
Example 2
For the reaction: A + B → C + B' (See illustration below):
Exercise
Exercise on Finding the Limiting Reactant
Check your answers here:
Solution to the Exercise on Finding the Limiting Reactant
Important Remarks 2:
- Finding the "right" limiting reactant is crucial in these problems, because it is used to calculate the # mol of products to be formed.
- If number of mol of both reactants are given, then finding the "right" limiting reactant is straightforward.
- If number of grams of both reactants are given, then converting these numbers to mol is essential to find the "right" limiting reactant.
- If either number of mol or number of grams of both reactants are given, then it is a "limiting reactant problem", even if finding the "right" limiting is not asked for.
In fact, in these cases, finding the "right" limiting reactant is a must to continue with the problem.
For more details, please contact me here.
