What is Empirical Formula from Analysis?
Introduction
Example 1
- 1 molecule of H2O contains 2 H atoms & 1 O atom.
- 1 mol of H2O contains 2 mol of H atoms and 1 mol of O atom.
- Hence, if the number of moles of the atoms are known, number of moles of their molecule can be determined.
The procedure is shown below:
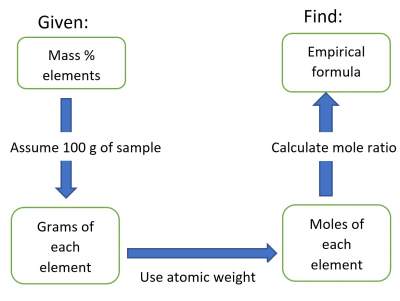
Example 2
Hg reacts with Cl forming a compound that is 73.9 % Hg and 26.1% Cl by mass.
Question 1: What is the empirical formula of the compound? (Note: Atomic weights of Hg and Cl are 200.6 and 35.5, respectively).
Solution:
- These two elements form ONE compound, whose formula is to be determined.
- The question is about the Empirical Formula; the simplest formula of this compound Procedure
- Assume 100 g sample, this contains: 73.9 g Hg & 26.1 g Cl
- Use the atomic weights of Hg and Cl, to calculate their number of moles: For Hg: # moles = (73.9 / 200.6) = 0.37
- For Cl: # moles = (26.1 / 35.5) = 0.74
- Calculate the molar ratio as follows: Hg : Cl (0.37 : 0.74)
- Divide all numbers by the smallest # of moles Hg : Cl (1 : 2)
Hence, the empirical formula is HgCl2
Question 2:
What is the relation between: a "Molecular Formula" and an "Empirical Formula"?
Answer:
Remember the following:
To find the empirical formula of the molecular formula, C6H12O6, we need to divide by the common number, which is 6 in this example, and we obain: CH2O
So, a molecular formula is an actual formula of the molecule, and the empirical formula is a simpler (abstracted) form.
Similarly, 180 g/mol (molecular formula weight), and then dividing it by 6 (the common number), we obtain 30 g/mol (empirical formula weight).
Hence, knowing the empirical formula, its weight and the molecular formula weight will let us determine the molecular formula.
For more details, please contact me here.
